Visual and overview of GISAID mutation counts data + covariates: early period
T. Bataillon
Last update: 2021-07-01 10:06:37
Last updated: 2021-07-01
Checks: 6 1
Knit directory: sars-cov2-gisaid/
This reproducible R Markdown analysis was created with workflowr (version 1.6.2). The Checks tab describes the reproducibility checks that were applied when the results were created. The Past versions tab lists the development history.
The R Markdown file has unstaged changes. To know which version of the R Markdown file created these results, you’ll want to first commit it to the Git repo. If you’re still working on the analysis, you can ignore this warning. When you’re finished, you can run wflow_publish to commit the R Markdown file and build the HTML.
Great job! The global environment was empty. Objects defined in the global environment can affect the analysis in your R Markdown file in unknown ways. For reproduciblity it’s best to always run the code in an empty environment.
The command set.seed(20210408) was run prior to running the code in the R Markdown file. Setting a seed ensures that any results that rely on randomness, e.g. subsampling or permutations, are reproducible.
Great job! Recording the operating system, R version, and package versions is critical for reproducibility.
Nice! There were no cached chunks for this analysis, so you can be confident that you successfully produced the results during this run.
Great job! Using relative paths to the files within your workflowr project makes it easier to run your code on other machines.
Great! You are using Git for version control. Tracking code development and connecting the code version to the results is critical for reproducibility.
The results in this page were generated with repository version 9c3698e. See the Past versions tab to see a history of the changes made to the R Markdown and HTML files.
Note that you need to be careful to ensure that all relevant files for the analysis have been committed to Git prior to generating the results (you can use wflow_publish or wflow_git_commit). workflowr only checks the R Markdown file, but you know if there are other scripts or data files that it depends on. Below is the status of the Git repository when the results were generated:
Ignored files:
Ignored: .DS_Store
Ignored: .Rhistory
Ignored: .Rproj.user/
Ignored: analysis/.Rhistory
Ignored: data/.DS_Store
Untracked files:
Untracked: analysis/1_working_with_the_data.snpeff.v2.gisaid_v2.0.rmd
Untracked: analysis/2_human_lineage_mutations.snpeff.v1.rmd
Untracked: analysis/hard_coded_params.R
Untracked: code/make_debug_miniature_dataset.R
Untracked: code/make_genome_positions.R
Untracked: data/2021-04-09/
Untracked: data/MutCounts_with_covariates.csv
Untracked: data/proteins_sarscov2_genome_positions.rds
Untracked: how_to_work_together.md
Untracked: output/README.html
Unstaged changes:
Modified: README.md
Modified: _workflowr.yml
Deleted: analysis/S0_creating_data_bywindows_lineages_OBSOLETE.Rmd
Modified: analysis/S0_creating_data_bywindows_lineages_byphase.Rmd
Modified: analysis/S1_adding_covariates_multiple_lineage_early_late.Rmd
Modified: analysis/S1_adding_covariates_multiple_lineage_period2.Rmd
Modified: analysis/S1_adding_covariates_multiple_lineage_period3.Rmd
Modified: analysis/S2_mutations_counts_covs_exploration_visuals.Rmd
Modified: analysis/S2_mutations_counts_covs_exploration_visuals_period2.Rmd
Modified: analysis/S2_mutations_counts_covs_exploration_visuals_period3.Rmd
Modified: analysis/S4_mutation_counts_models_period1.Rmd
Modified: analysis/S4_mutation_counts_models_period2.Rmd
Modified: analysis/S4_mutation_counts_models_period3.Rmd
Modified: analysis/_site.yml
Modified: analysis/about.Rmd
Modified: analysis/index.Rmd
Modified: code/README.md
Modified: data/README.md
Modified: output/README.md
Note that any generated files, e.g. HTML, png, CSS, etc., are not included in this status report because it is ok for generated content to have uncommitted changes.
These are the previous versions of the repository in which changes were made to the R Markdown (analysis/S2_mutations_counts_covs_exploration_visuals_period3.Rmd) and HTML (docs/S2_mutations_counts_covs_exploration_visuals_period3.html) files. If you’ve configured a remote Git repository (see ?wflow_git_remote), click on the hyperlinks in the table below to view the files as they were in that past version.
| File | Version | Author | Date | Message |
|---|---|---|---|---|
| Rmd | 9c3698e | tbata | 2021-06-30 | Added 100 nt windows for 3 periods on downsized 300k data. and update of all analysis |
# data_path <- "data/2021-06-18/"
source("analysis/hard_coded_params.R")Using data version: data/2021-06-18/
Loading data from bins and covariates
suppressPackageStartupMessages(library(tidyverse)) # to avoid messy
library(ggsci)
library(ggrepel)
library(cowplot)
data100_annotated<- readRDS( "output/data_100pb_pangolin_period3_withCovs.rds")
dim(data100_annotated)[1] 4298 24names(data100_annotated) [1] "gene_name" "gene_bin" "type"
[4] "W.R.dS" "W.G.dS" "R.G.dS"
[7] "ii.CD4" "ii.CD8" "ii.CD4.max"
[10] "ii.CD8.max" "ENC" "ENC_STD"
[13] "GC" "CAI" "CAI_STD"
[16] "PPI" "GO" "Prot.Dom"
[19] "pangolin_lineage" "gene" "unique_mutations"
[22] "max_genomes_mutated" "window_start" "window_start_gis" table(data100_annotated$gene_name)
E endoRNAse exonuclease helicase
42 154 224 266
leader M methyltransferase N
84 98 126 182
nsp10 nsp11 nsp2 nsp3
70 14 280 826
nsp4 nsp6 nsp7 nsp8
224 126 42 84
nsp9 orf10 orf3a orf6
56 28 126 28
orf7a orf7b orf8 proteinase
56 28 56 140
RDRp S
392 546 table(data100_annotated$gene)
E endoRNAse exonuclease helicase
31 118 182 205
leader M methyltransferase N
82 74 96 174
nsp10 nsp11 nsp2 nsp3
66 11 271 779
nsp4 nsp6 nsp7 nsp8
197 117 39 74
nsp9 orf10 orf3a orf6
51 22 116 25
orf7a orf7b orf8 proteinase
50 22 52 128
RDRp S
317 460 Overview dataset
data100_annotated %>%
select(gene_name, gene_bin, type, unique_mutations, W.G.dS) %>%
slice(400:410) %>%
knitr::kable(digits = 2)| gene_name | gene_bin | type | unique_mutations | W.G.dS |
|---|---|---|---|---|
| nsp8 | 3 | S | 8 | 0.39 |
| nsp8 | 4 | NS | 6 | 0.45 |
| nsp8 | 4 | S | 7 | 0.45 |
| nsp8 | 5 | NS | 9 | 0.52 |
| nsp8 | 5 | S | 7 | 0.52 |
| nsp8 | 6 | S | 5 | 0.57 |
| nsp8 | 6 | NS | 9 | 0.57 |
| nsp9 | 1 | NS | 8 | 0.65 |
| nsp9 | 1 | S | 7 | 0.65 |
| nsp9 | 2 | S | 9 | 0.75 |
| nsp9 | 2 | NS | 8 | 0.75 |
Overview counts
checking distributions
summary(data100_annotated$unique_mutations) Min. 1st Qu. Median Mean 3rd Qu. Max.
0.00 1.00 4.00 8.27 10.00 79.00 mutations along the genome
ggplot(data100_annotated, aes(x=window_start_gis, y=unique_mutations, size=max_genomes_mutated, color=type)) +
geom_point() +
ylim(c(0,NA)) +
xlim(c(0,NA)) +
xlab("Genome position") +
ylab("Number of unique mutations in 100bp") +
scale_color_aaas(name="") +
scale_size("Max genomes mutated", range=c(1,5)) +
theme(legend.position = c(0.5,1),legend.justification = c(0.5,1))+
# theme(legend.box.background = element_rect(fill="#F0F0F0")) +
NULL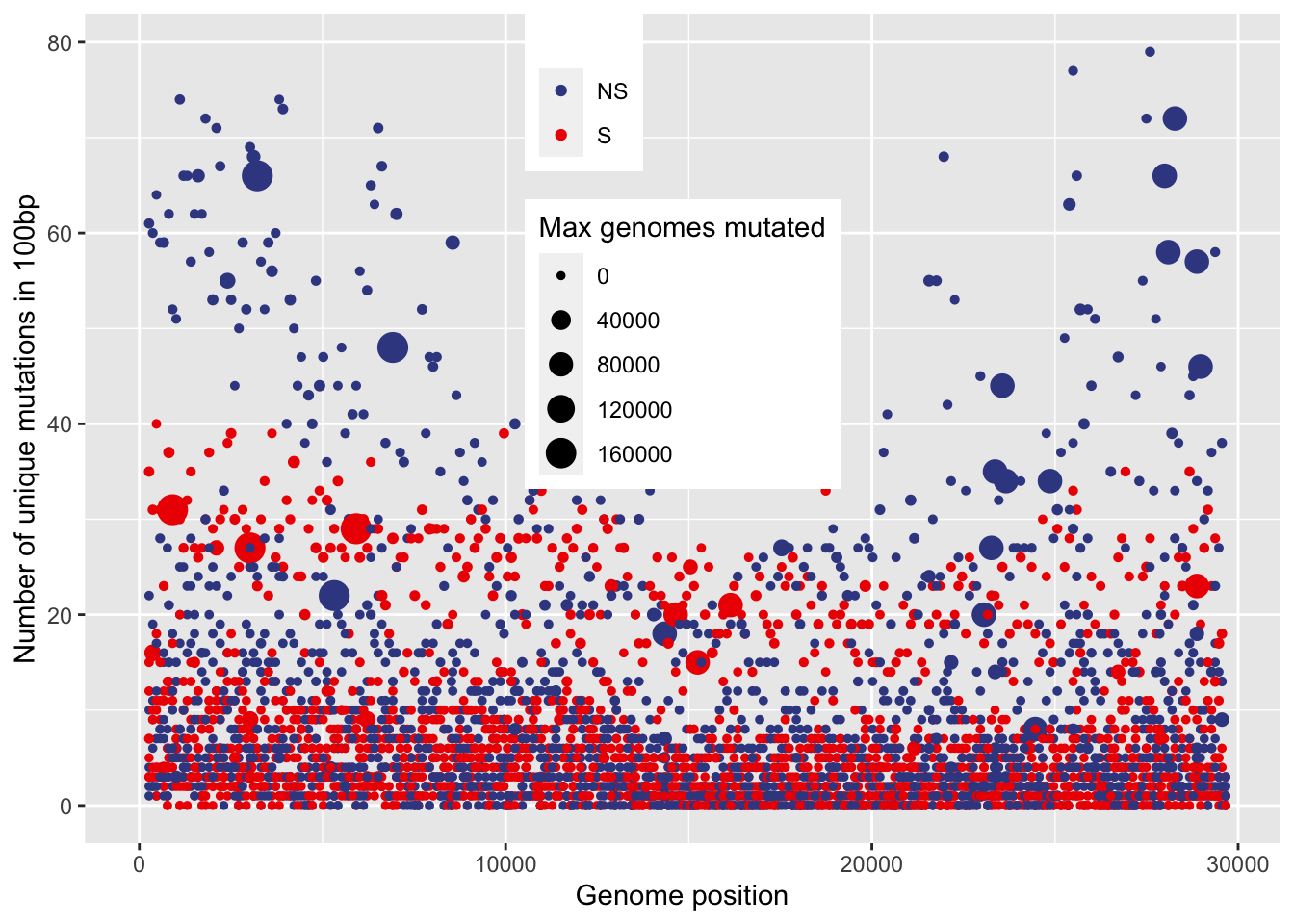
histogram of counts
ggplot(data100_annotated, aes(x=unique_mutations, fill=type)) +
geom_histogram() +
ylim(c(0,NA)) +
xlim(c(0,NA)) +
ylab("Number of windows") +
xlab("Number of unique mutations in 100bp") +
scale_fill_aaas(name="") +
facet_wrap(~ pangolin_lineage + type, ncol = 2, scales = "free_y")+
# theme(legend.position = c(0.5,1),legend.justification = c(0.5,1))+
# theme(legend.box.background = element_rect(fill="#F0F0F0")) +
NULL`stat_bin()` using `bins = 30`. Pick better value with `binwidth`.Warning: Removed 14 rows containing missing values (geom_bar).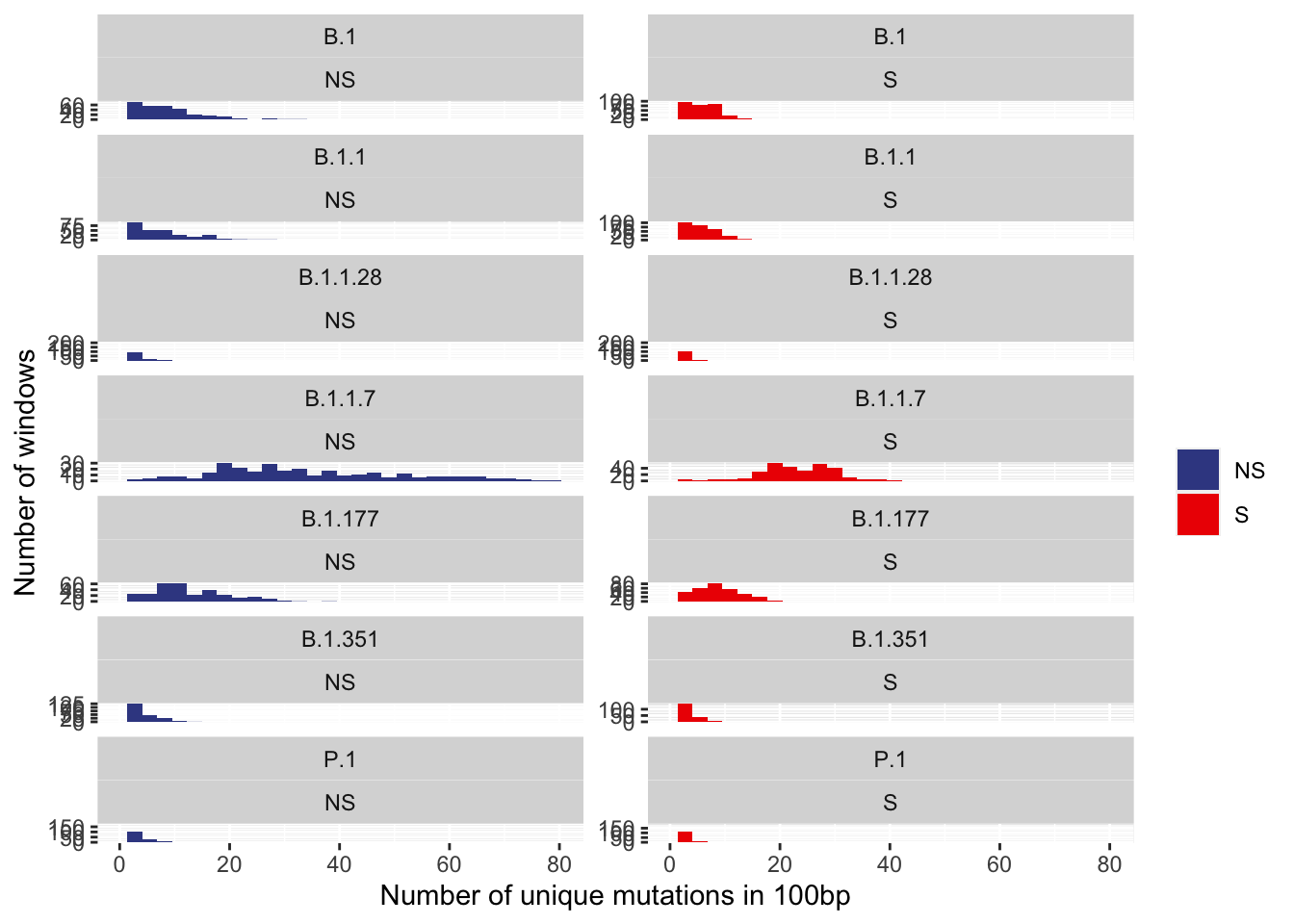
Counts of mutations versus immunogenicity index
data100_annotated %>%
filter(gene %in% c("N","M","S")) %>%
filter(type == "NS") %>%
ggplot(aes(x=ii.CD8.max, y=unique_mutations, size=max_genomes_mutated, color=gene_name)) +
geom_point() +
geom_smooth(method = "lm", se=T, color= "black")+
ylim(c(0,NA)) +
xlim(c(0,NA)) +
xlab("ii.CD8max") +
ylab("Number of non syn mutations in 100bp") +
scale_color_aaas(name="") +
scale_size("Max genomes mutated", range=c(0.8,3)) +
facet_wrap(~ pangolin_lineage , ncol = 2)+
# theme(legend.position = "none")+
# theme(legend.position = c(0.5,1),legend.justification = c(0.5,1))+
# theme(legend.box.background = element_rect(fill="#F0F0F0")) +
NULL`geom_smooth()` using formula 'y ~ x'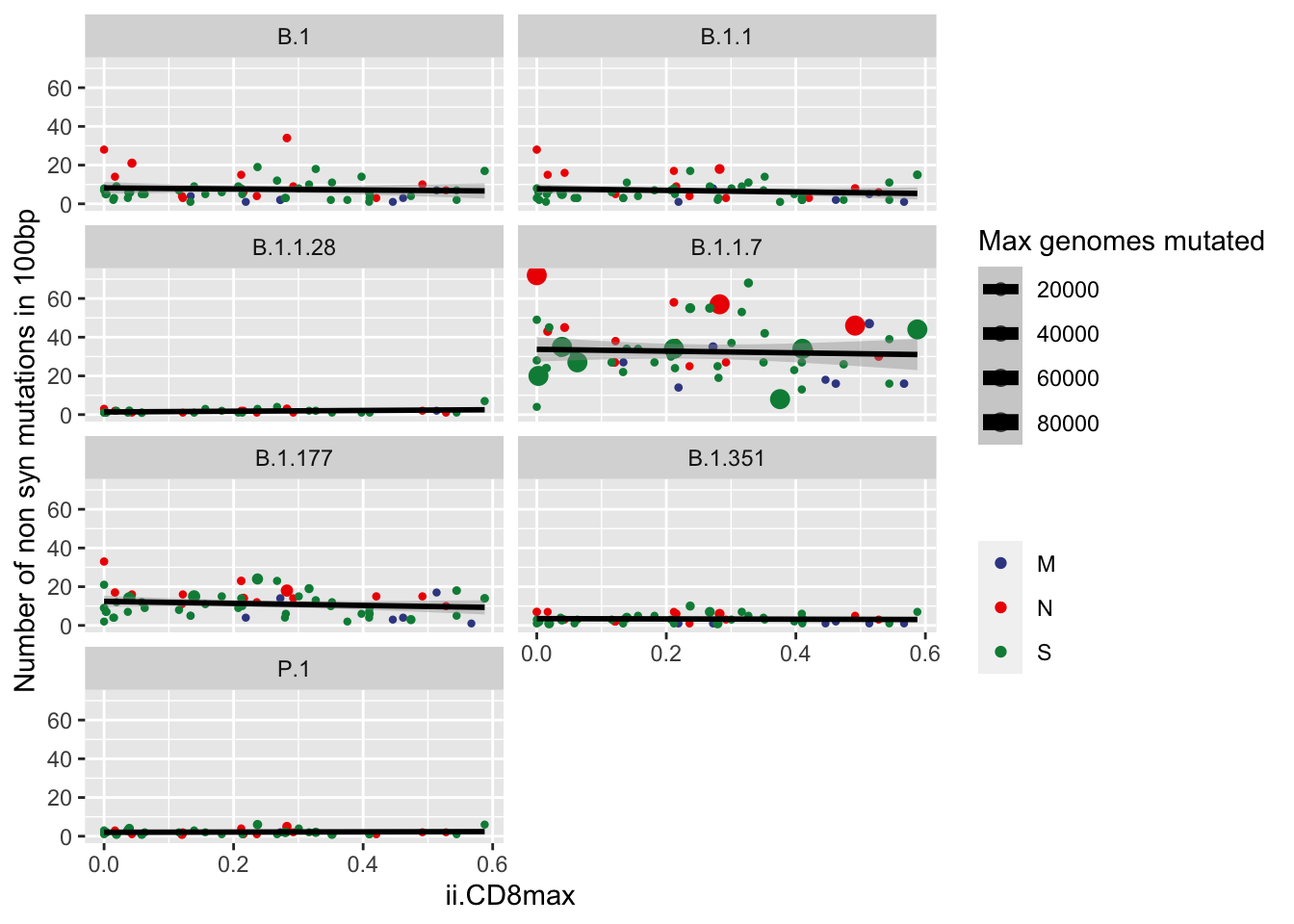
dS versus unique mutations
names(data100_annotated) [1] "gene_name" "gene_bin" "type"
[4] "W.R.dS" "W.G.dS" "R.G.dS"
[7] "ii.CD4" "ii.CD8" "ii.CD4.max"
[10] "ii.CD8.max" "ENC" "ENC_STD"
[13] "GC" "CAI" "CAI_STD"
[16] "PPI" "GO" "Prot.Dom"
[19] "pangolin_lineage" "gene" "unique_mutations"
[22] "max_genomes_mutated" "window_start" "window_start_gis" data100_annotated %>%
filter(type == "NS") %>%
ggplot(aes(x= log10(1+W.G.dS) , y=unique_mutations, size=max_genomes_mutated, color=gene_name)) +
geom_jitter(height = 0.2) +
ylim(c(0,NA)) +
xlim(c(0,NA)) +
xlab("log10(dS) ") +
ylab("Number of non syn mutations in 100bp") +
scale_size("Max genomes mutated", range=c(0.8,3)) +
scale_color_viridis_d(direction = -1)+
facet_wrap(~ pangolin_lineage , ncol = 1, scales = "free_y")+
geom_smooth(method = "loess", aes(color=NULL), color= "black", size=0.5)+
# theme(legend.position = "none")+
# theme(legend.position = c(0.5,1),legend.justification = c(0.5,1))+
# theme(legend.box.background = element_rect(fill="#F0F0F0")) +
NULL`geom_smooth()` using formula 'y ~ x'Warning: Removed 146 rows containing missing values (geom_point).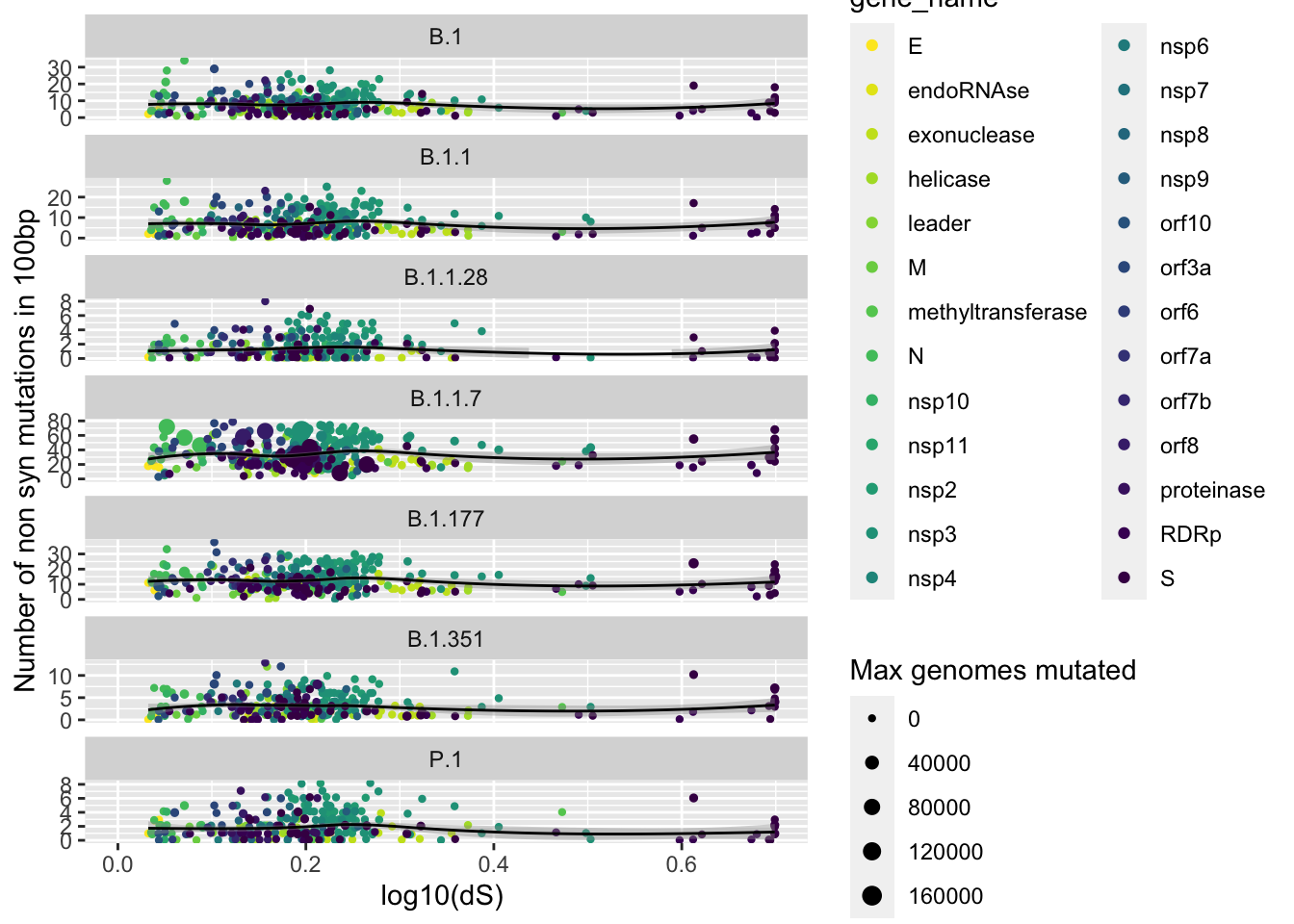
sessionInfo()R version 4.0.2 (2020-06-22)
Platform: x86_64-apple-darwin17.0 (64-bit)
Running under: macOS Catalina 10.15.7
Matrix products: default
BLAS: /Library/Frameworks/R.framework/Versions/4.0/Resources/lib/libRblas.dylib
LAPACK: /Library/Frameworks/R.framework/Versions/4.0/Resources/lib/libRlapack.dylib
locale:
[1] en_US.UTF-8/en_US.UTF-8/en_US.UTF-8/C/en_US.UTF-8/en_US.UTF-8
attached base packages:
[1] stats graphics grDevices utils datasets methods base
other attached packages:
[1] cowplot_1.1.0 ggrepel_0.9.1 ggsci_2.9 forcats_0.5.0
[5] stringr_1.4.0 dplyr_1.0.2 purrr_0.3.4 readr_1.3.1
[9] tidyr_1.1.1 tibble_3.0.3 ggplot2_3.3.2 tidyverse_1.3.0
[13] workflowr_1.6.2
loaded via a namespace (and not attached):
[1] Rcpp_1.0.5 lattice_0.20-41 lubridate_1.7.9 assertthat_0.2.1
[5] rprojroot_2.0.2 digest_0.6.25 R6_2.4.1 cellranger_1.1.0
[9] backports_1.1.9 reprex_0.3.0 evaluate_0.14 httr_1.4.2
[13] highr_0.8 pillar_1.4.6 rlang_0.4.7 readxl_1.3.1
[17] rstudioapi_0.11 whisker_0.4 blob_1.2.1 Matrix_1.2-18
[21] rmarkdown_2.3 splines_4.0.2 labeling_0.3 munsell_0.5.0
[25] broom_0.7.0 compiler_4.0.2 httpuv_1.5.4 modelr_0.1.8
[29] xfun_0.16 pkgconfig_2.0.3 mgcv_1.8-31 htmltools_0.5.0
[33] tidyselect_1.1.0 viridisLite_0.4.0 fansi_0.4.1 crayon_1.3.4
[37] dbplyr_1.4.4 withr_2.2.0 later_1.1.0.1 grid_4.0.2
[41] nlme_3.1-148 jsonlite_1.7.1 gtable_0.3.0 lifecycle_0.2.0
[45] DBI_1.1.0 git2r_0.27.1 magrittr_1.5 scales_1.1.1
[49] cli_2.0.2 stringi_1.4.6 farver_2.0.3 fs_1.5.0
[53] promises_1.1.1 xml2_1.3.2 ellipsis_0.3.1 generics_0.0.2
[57] vctrs_0.3.2 tools_4.0.2 glue_1.4.1 hms_0.5.3
[61] yaml_2.2.1 colorspace_1.4-1 rvest_0.3.6 knitr_1.29
[65] haven_2.3.1